Introduction
Every reliable water disinfection process operates through oxidation, a mechanism that strips electrons from pathogen cellular structures to destroy them. Historically, facilities relied on standard dosing protocols for the chemicals used in water treatment, and these protocols assume that source water chemistry remains relatively stable throughout the year. However, dissolved organic matter in source water frequently competes with pathogens for these exact same oxidants, and when organic load rises above baseline assumptions, correctly dosed treatment programs fail because the chemical is consumed before it reaches its target.
This dynamic creates a critical chemistry competition problem rather than a standard contamination issue. Research shows that extreme rainfall events can increase raw water dissolved organic carbon from 3.9 to 14.4 mg/L, pushing conventional systems well beyond their design thresholds. The selection of correct treatment chemistry requires accounting for this organic load variability, especially when organic content fluctuates with seasons, weather patterns, and upstream activity.
How Chemicals Used in Water Treatment Operate at Molecular Level
Every chemical disinfectant destroys pathogens through oxidation, which strips electrons from the molecular structures that hold a pathogen together and breaks apart cell walls, disrupts membranes, and deactivates enzymes a microorganism needs to survive. The chemicals used in water treatment function as oxidants and their differences lie not in their objective but in how many oxidation pathways they possess and how quickly competing molecules exhaust their capacity. The concept of water quality enhancement through filtration and oxidation depends entirely on whether enough of that oxidation budget survives long enough to reach microbial targets.
Oxidation capacity operates like a finite budget. A precise dose of any oxidant carries a fixed number of reactive molecules into the water, and each molecule can participate in only one chemical reduction reaction before it is spent. If those molecules react with something other than the intended pathogen targets first, the budget is gone and pathogens remain intact.
Competition of Organic Molecules Against Pathogens
Dissolved organic compounds in source water react with and consume the same oxidant molecules that destroy bacteria, viruses, and protozoa. This is not contamination in the traditional sense but a measurable chemistry competition where organic matter outcompetes pathogens for available disinfectant. Source water carries organic compounds from humic and fulvic acids from decomposing vegetation, residual agricultural chemicals, and dissolved plant matter from seasonal runoff.
Chlorine demand shows a strong positive correlation with dissolved organic carbon concentration, and when organic load rises, the chemicals used in water treatment undergo reduction reactions with dissolved organics before they ever contact a single pathogen. The water passes through the treatment process and appears treated, but it is not disinfected. This competition problem compounds the limitations that already exist with conventional oxidants — even under ideal low-organic conditions, chlorine shows limited effectiveness against Cryptosporidium and Giardia.
When organic load consumes available chlorine before it contacts pathogens, those already limited effectiveness rates drop further. This failure fundamentally breaks down the disinfection process itself rather than creating a minor operational nuisance. Chemical oxidation remains the primary disinfection mechanism for most treatment systems, which makes this vulnerability a structural problem rather than an edge case.
Environments Vulnerable to Organic Load Fluctuations
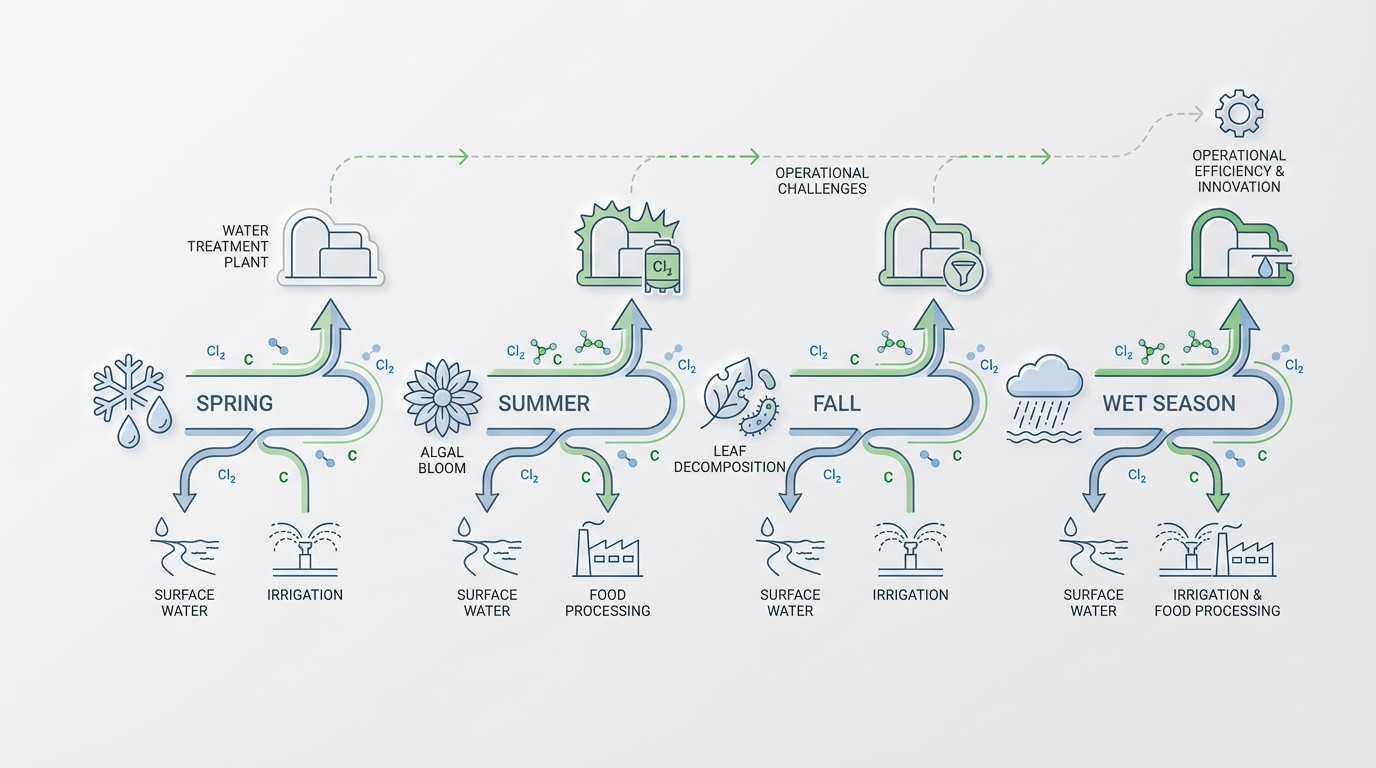
Surface water sources face the widest swings in dissolved organic content, and those swings follow structured seasonal patterns. Spring snowmelt flushes accumulated organic matter from fields and wetlands, summer brings algal blooms, and fall delivers leaf litter and crop residue decomposition. Each season creates a predictable window during which source water organic load rises and conventional treatment chemistry becomes less reliable.
Research confirms that wet seasons with high rainfall consistently show elevated chlorine consumption compared to dry seasons, driven directly by rising dissolved organic matter concentrations. Irrigation systems draw from these surface sources and absorb the full impact of these fluctuations. Food processing facilities face the same challenge, particularly where water treatment in the food industry must meet strict pathogen control standards regardless of incoming water quality.
In food processing wash operations, sugars, organic acids, and proteins released from produce accumulate rapidly in recirculated water and deplete available chlorine sanitizer. These environments do not experience organic load spikes as surprises — the spikes create foreseeable chemistry challenges tied to weather, season, and upstream land use. They drive treatment system design decisions rather than trigger reactive troubleshooting after disinfection fails.
Inherent Vulnerabilities of Single Oxidation Pathway
Chlorine exists as hypochlorous acid or hypochlorite ion and reacts with whatever oxidizable material it contacts first, without distinguishing between a dissolved humic acid molecule and a bacterial cell wall. Both present electron-rich molecular structures, and chlorine attacks whichever it encounters on a first-contact basis. The chemicals used in water treatment that rely on this single pathway carry no backup mechanism. Once available chlorine molecules react with organic compounds, the disinfection budget is spent.
No secondary oxidation chemistry activates to continue pathogen destruction. An analysis of how residential wastewater disinfection differs in practice illustrates the same principle at a smaller scale. One pathway leaves no margin for competing reactions, while two pathways build redundancy into the chemistry itself.
Direct Oxidation Limitations
In low-organic-load water, direct oxidation works well and most chlorine molecules reach pathogen targets with sufficient concentration to achieve cell destruction. The fundamental problem emerges when organic load rises, because each milligram of dissolved organic carbon that enters the treatment zone triggers reduction reactions that consume chlorine molecules before those molecules contact a single bacterium or virus. At elevated organic concentrations, these reactions alone can exhaust the entire chlorine dose, leaving zero residual available for disinfection.
Ozone Systems for Byproduct Control
Chlorine reacts with organic matter to produce trihalomethanes and haloacetic acids, both classified as disinfection byproducts, and higher chlorine doses under high-organic conditions generate proportionally more of these byproducts. A rainfall event that increased raw water dissolved organic carbon from 3.9 to 14.4 mg/L produced disinfection byproduct concentrations that rose 1.5 to 2-fold above baseline levels. Treatment systems cannot achieve adequate disinfection and acceptable byproduct levels simultaneously when organic load rises and chlorine acts as the only available oxidant.
Half-Life and Stability Constraints
A switch from free chlorine to chloramines does not resolve the organic competition problem. Chloramines maintain a longer half-life in distribution systems than free chlorine, which means they persist longer, but persistence and pathway redundancy are different properties. Greater stability simply slows the rate at which organic compounds exhaust the available oxidant. Chloramines delay the same failure rather than prevent it.
Why Dual Oxidation Pathways Ensure Reliability
Ozone operates through two distinct and simultaneous oxidation pathways. The first is direct molecular ozone attack, where ozone molecules selectively target specific chemical bonds in both organic compounds and pathogen cell structures. The second is hydroxyl radical generation — as ozone decomposes in water, it produces hydroxyl radicals with an oxidation potential of 2.80 volts, compared to 2.07 volts for ozone and 1.36 volts for chlorine.
Hydroxyl radicals react with nearly any organic compound at near diffusion-limited rates, and ozone-based advanced oxidation combines direct molecular attack with indirect hydroxyl radical oxidation as two parallel and complementary pathways. When dissolved organic matter consumes a portion of the direct ozone molecules, hydroxyl radical production continues, and when organic compounds react with hydroxyl radicals, direct ozone molecules remain available to attack pathogens. Neither pathway depends on the other to function, and each compensates when the competing pathway is partially consumed.
This creates a strong disinfection architecture rather than a fragile single point of failure. For facilities that manage surface water contamination in the MENA region and similar variable-quality sources, ozone solutions provide a secure oxidation framework that maintains pathogen destruction even when organic load absorbs a significant fraction of the total oxidant dose. The system bends under organic demand pressure and does not break.
Treatment Chemistry and Source Water Realities
The choice of the right treatment chemistry starts with an honest assessment of source water organic load variability rather than a product catalog. Facilities that draw from deep wells or protected groundwater typically manage stable, low-organic-load water where chlorination performs within its design parameters. Chemical reduction reactions between chlorine and competing organic matter stay minimal, and adequate disinfectant residual reaches pathogens consistently.
Facilities that draw from surface water intakes, shallow wells, or seasonal water sources face a different reality. Climate-driven increases in natural organic matter are placing sustained upward pressure on dissolved organic carbon concentrations in surface water sources globally, meaning treatment programs calibrated to historical baseline conditions are increasingly operating outside their design envelope. For these sources, aligning oxidation mechanism to the expected range of organic fluctuation produces more reliable outcomes than calibrating to a single baseline measurement.
A practical framework for this evaluation considers the following factors:
-
Sources with dissolved organic carbon consistently below 4 mg/L can sustain chlorination at standard doses without significant organic competition.
-
Sources with dissolved organic carbon ranging between 4 and 10 mg/L require either continuous monitoring with adaptive dosing or a shift to ozone solutions that tolerate the organic demand without byproduct escalation.
-
Sources with dissolved organic carbon exceeding 10 mg/L during seasonal or storm-driven events need dual-pathway oxidation chemistry to maintain disinfection through peak organic load windows.
Treatment chemistry and source water chemistry must align. When they do not, the gap between designed performance and actual performance grows with every organic load spike.
Conclusion
Dissolved organic load dictates whether oxidation-based disinfection programs achieve their fundamental objectives. Facilities that manage variable source water face unacceptable vulnerabilities when they rely on a single oxidation pathway, and as environmental conditions continue to drive wider fluctuations in source water quality, resilient dual-pathway systems like the ozone method of water treatment will become the standard for modern water management.
Agritopia supplies ozone water treatment equipment designed to address variable organic load conditions in agricultural and food processing operations across the MENA region. Contact us to find out which ozone treatment configuration fits your facility's specific source water profile and contamination challenges.






