Introduction
Agricultural, institutional, and food processing facilities rely on consistent wastewater treatment processes to maintain regulatory compliance. Engineers typically calibrate fixed chlorine dosing systems for average organic loads. However, these systems systematically underperform during seasonal production peaks.
Surges of organic matter enter the facility and consume available chlorine before the chemical neutralizes target pathogens. This failure represents an inherent chemical limitation that intensifies when treatment reliability matters most. Operations face growing pressure to adapt. The United Nations issued a water bankruptcy warning because 15 of the world's 20 most water-scarce countries sit in the Middle East and North Africa region.
Standard dosing protocols leave facilities exposed to significant compliance risks during high-load periods. Ozone-based treatment solves this problem through fundamentally different chemistry. Ozone utilizes two distinct oxidation pathways and maintains disinfection efficacy regardless of fluctuating organic loads. This chemical distinction helps facilities protect infrastructure and avoid unexpected failures throughout the year.
Calibration Limitations of Fixed Chlorine Protocol
Most facilities calibrate chlorine protocols around average influent organic loads rather than the dramatic surges that arrive during harvest cycles, industrial production spikes, and peak-season processing runoff. These facilities operate systems designed for a narrow band of expected conditions, and the math works well enough during steady-state months. However, these operations face problems when incoming chemical oxygen demand and biochemical oxygen demand spike beyond those design assumptions.
This structural mismatch occurs between system calibration and how wastewater treatment processes actually behave across an annual production calendar. The chlorine volume that kept effluent compliant in February fails to handle what a facility produces in September. Fixed-dose systems enter a predictable failure mode during the exact periods when treatment reliability matters most.
Seasonal variability also disrupts downstream crop protection systems that depend on treated water quality. When facilities follow standard protocols and fail to account for peak loads, treatment optimisation becomes reactive rather than preventive. This approach widens the gap between design conditions and field conditions every production season, and this gap exposes the fundamental chemical weaknesses of standard treatment methods.
Chemical Mechanics of Chlorine Demand
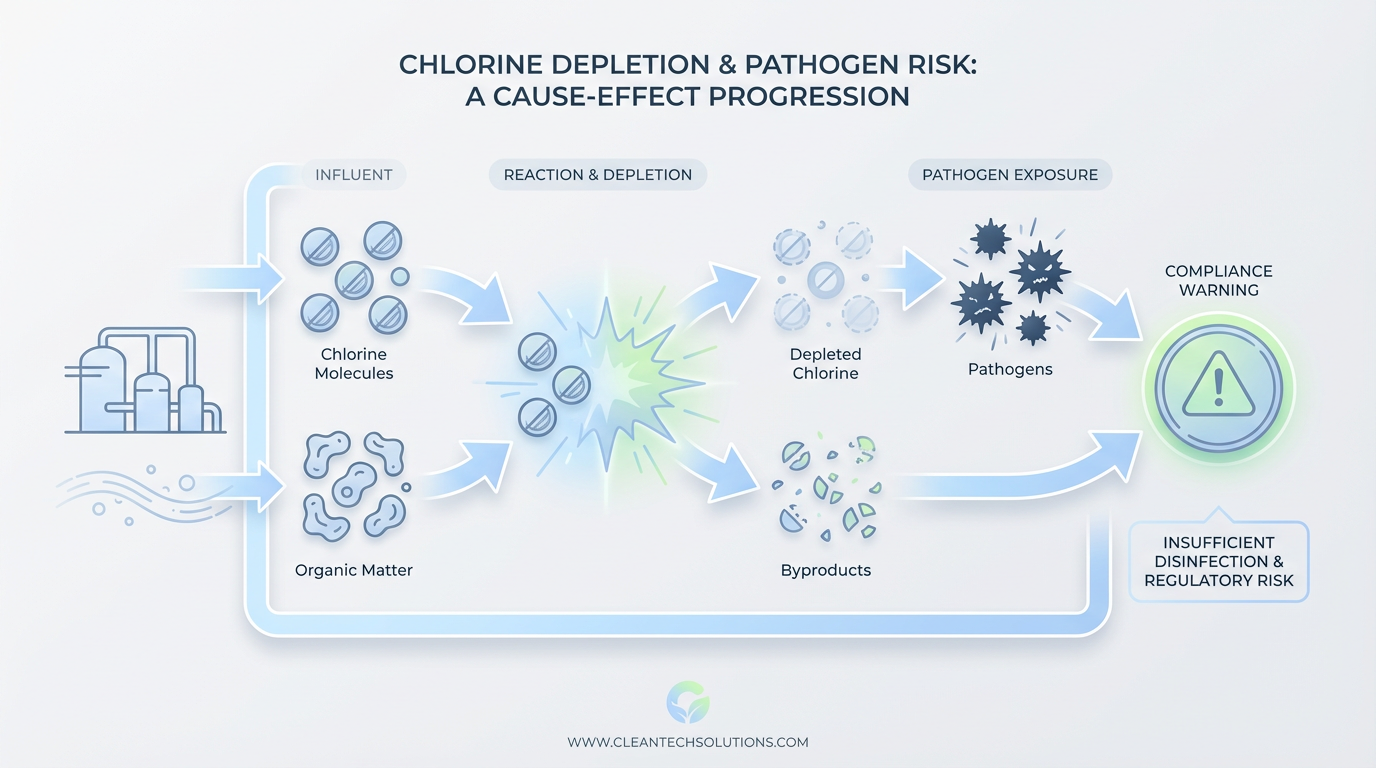
Chlorine serves as a selective and relatively weak oxidizer, and organic matter exploits that weakness during seasonal peaks. When proteins, sugars, fats, and phenolic compounds flood a treatment system, chlorine reacts preferentially with these dissolved and particulate organics rather than with target pathogens. Chemists call this phenomenon chlorine demand, and it depletes available free chlorine before disinfection occurs.
The chemistry compounds the problem further. As chlorine reacts with abundant organics, it generates regulated disinfection byproducts, such as trihalomethanes and haloacetic acids. These secondary compounds create a dual compliance failure because the system fails to disinfect and produces harmful byproducts at elevated concentrations. The expectation that a standard dose will maintain stability across variable influent conditions contradicts what the chemistry actually does under load.
This chemical limitation matters for wastewater treatment processes that feed residential wastewater disinfection and broader reuse systems alike. A treatment train designed to protect public health fails to fulfill that role when the effluent stream consumes the primary disinfectant before it reaches pathogens, and this rapid consumption happens because wastewater properties constantly change.
Compositional Shifts During Seasonal Cycles
Agricultural and industrial wastewater is not static. Its composition shifts on a schedule that harvest calendars, processing lines, and cleaning cycles dictate, and each shift brings a distinct organic load profile that fixed-dose systems fail to handle. The reliability of any treatment system depends on how well it accommodates these predictable yet dramatic changes.
Specific operations generate specific challenges across the year:
-
Fruit and vegetable processing creates sugar-rich wastewater with elevated soluble chemical oxygen demand from washing, peeling, and blanching.
-
Dairy operations generate whey-laden effluent that contains lactose, casein, and milk solids that spike during peak production months.
-
Textile and paper manufacturing plants discharge chemical dyes and bleaching agents that complicate standard treatment protocols.
-
Crop washing introduces sediment, vegetative debris, and pesticide residues that vary by crop type and field conditions.
-
Cleaning cycles between production runs add detergents and sanitizers that alter wastewater chemistry unpredictably.
These compositional shifts follow seasonal patterns closely enough to map, yet wastewater treatment processes calibrated to annual averages treat every influent stream as if it were the same. Downstream reuse systems inherit whatever the treatment train failed to handle. Facilities face a growing gap between design assumptions and operational reality with each passing season because they lack protection against these foreseeable surges, and this vulnerability requires an alternative chemical approach like ozone.
Direct Molecular Attack in Effluent Treatment
Ozone's first oxidation pathway operates through direct molecular contact. Ozone molecules selectively target electron-rich functional groups on organic contaminants, such as aromatic rings and unsaturated carbon-carbon double bonds. This selectivity gives the pathway a form of chemical assurance because it attacks the structural features most commonly found in the complex organics that dominate agricultural and industrial effluent.
When ozone contacts these functional groups, it fragments large organic molecules into smaller and more biodegradable compounds, such as organic acids, aldehydes, and ketones. Downstream biological treatment stages metabolize these breakdown products much easier. The fragmentation process reduces contaminant concentration and changes the character of the wastewater itself, and it converts recalcitrant compounds into forms that biological processes can finish removing.
This distinction separates ozone from chlorine at a fundamental level. Chlorine becomes consumed through reactions with organic matter, and this dynamic leaves pathogens safe behind that same organic load. Ozone's direct pathway constructively dismantles the organic barrier, and the destruction of complex organics functions as a productive treatment step rather than a waste of chemical reagent. Each molecule of ozone spent on organic fragmentation improves the treatability of the entire effluent stream and preserves the system's disinfection capacity.
Indirect Hydroxyl Radical Generation
Ozone's second oxidation pathway emerges when ozone molecules decompose in water and produce hydroxyl radicals (·OH). These radicals rank among the most reactive oxidizing species in water treatment, and they behave differently from the direct pathway because they do not discriminate. Hydroxyl radicals attack virtually all organic structures at near-diffusion-limited reaction rates, and they react with contaminants almost as fast as they encounter them.
This non-selective power achieves what selective oxidants cannot. Hydroxyl radicals do not fragment specific molecular bonds but drive complete mineralization. They convert complex organic pollutants into stable inorganic end products, such as water, carbon dioxide, and mineral salts. No regulated disinfection byproducts form during this process, and this removes a secondary compliance risk that chlorine-based treatment cannot avoid.
The certainty that this pathway provides matters for treatment optimisation during peak-load periods. When organic concentrations surge, hydroxyl radical generation increases proportionally because more ozone decomposition occurs in the presence of higher organic loads. Facilities that manage surface water contamination treatment alongside process wastewater benefit from this self-reinforcing chemistry, and this chemistry intensifies precisely when contaminant loads demand the most aggressive oxidation response that single-mechanism systems cannot provide.
Dual Oxidation Pathways Ensure Reliability
Chlorine operates through a single oxidation mechanism. When organic matter overwhelms that mechanism, the system lacks a fallback. Ozone's two pathways function simultaneously and complement each other, and this distinction explains why ozone adapts where chlorine depletes.
The direct pathway fragments complex organic molecules into smaller, biodegradable compounds. The indirect hydroxyl radical pathway mineralizes residual organics and attacks pathogens non-selectively. These two mechanisms share the oxidation workload across the full spectrum of contaminants that exist in the effluent stream at any given moment.
During seasonal peaks, both pathways continue operating. Higher organic loads do not exhaust ozone's treatment capacity the way they exhaust chlorine's available free residual. Instead, the chemistry self-adjusts because more organics generate more hydroxyl radicals through increased ozone decomposition, and the direct pathway handles the structural dismantling of large molecules that would otherwise shield pathogens.
Structural Fragmentation Benefits
Direct oxidation targets the electron-rich functional groups that make complex organic molecules resistant to conventional disinfection. Aromatic rings, unsaturated bonds, and branched carbon chains all provide structural protection for microorganisms that hide inside organic matter. Ozone cleaves these structures at their most vulnerable points.
The fragments that remain after direct oxidation include organic acids, aldehydes, and short-chain ketones, and they are smaller, simpler, and far more accessible to both downstream biological processes and the hydroxyl radical pathway. This fragmentation strips away the organic barrier that fixed-dose chlorine systems cannot penetrate during high-load conditions. Treatment optimisation depends on this sequential breakdown because it converts the organic load from a liability into a series of progressively treatable compounds instead of a wall that blocks disinfection entirely.
Complete Mineralization Benefits
The hydroxyl radical pathway finishes the work that direct oxidation starts. These radicals attack residual organic compounds without preference and oxidize whatever molecular structures remain after initial fragmentation. The end products are carbon dioxide, water, and inorganic salts rather than the trihalomethanes and haloacetic acids that chlorine produces under identical high-load conditions.
This absence of regulated byproducts during peak organic loads addresses the secondary failure that chlorine systems face. Chlorine does not just fail to disinfect when organics surge, but it also actively generates harmful compounds. Hydroxyl radical mineralization eliminates both problems in a single step. The reliability of this pathway during variable load conditions removes the compliance risk that compounds every time seasonal influent characteristics shift beyond design parameters, and this reliability helps operations meet strict environmental rules.
Regulatory Pressure and Compliance Gaps
Environmental discharge standards do not include seasonal exemptions. Facilities must maintain consistent effluent quality year-round regardless of how dramatically influent composition changes, and that regulatory reality exposes a growing gap between what fixed chlorine systems deliver and what compliance frameworks demand.
The security of a facility's operating permit depends on meeting carbonaceous Biochemical Oxygen Demand (BOD) thresholds below 25 mg/L and total suspended solids below 25 mg/L during every discharge event, not just during average-load months. When chlorine-based systems fail during production peaks, facilities face violation notices during the exact periods when production revenue peaks. Emerging contaminant regulations target per- and polyfluoroalkyl substances (PFAS), pharmaceutical residues, and persistent organic pollutants, and they push treatment demands further beyond chlorine's oxidation capacity.
Market data reflects this intensifying pressure. The global wastewater treatment market is projected to reach $127.54 billion by 2035, and this trajectory follows tightening discharge standards and the expansion of reuse systems that demand higher effluent quality than simple discharge permits require. Three regulatory trends accelerate this demand.
-
Regulators adopt stricter effluent limits for nutrient discharge, such as nitrogen and phosphorus, across industrial processing jurisdictions.
-
Authorities enforce maximum residual chlorine limits below 0.02 mg/L in receiving waters, and this penalizes facilities that increase chlorine dosing to compensate for seasonal organic loads.
-
Agencies expand monitoring requirements to include continuous compliance verification rather than periodic grab sampling.
Adaptive Design for Seasonal Realities
Engineers who design for average conditions and hope peaks remain manageable do not use a proper treatment strategy. This approach relies on an assumption that seasonal organic load data contradicts every year. Facilities that engineer their wastewater treatment processes around peak-load conditions as the baseline establish operational stability instead of chasing compliance failures after they occur.
Ozone-based systems integrate with real-time influent monitoring and automated mass transfer controls, and they provide the adaptive capacity that seasonal industrial operations require. Properly engineered ozone systems focus on mass transfer efficiency rather than oversized generator capacity, and integration with digital control platforms enables dynamic response to fluctuating influent characteristics. Operators pair these systems with water quality enhancement techniques, such as membrane polishing and catalytic stages, to create treatment trains that adjust to compositional shifts instead of breaking under them.
The economic argument for adaptive design extends beyond compliance. An April 2026 Cornell University study found that U.S. animal and human waste could meet 102% of nitrogen and 50% of phosphorus industrial needs, and this recovered resource is valued at $5.7 billion annually. Treated industrial wastewater becomes a nutrient recovery asset when the treatment infrastructure handles variable loads without sacrificing effluent quality.
Trust in reuse systems depends on the upstream treatment train's ability to deliver consistent output across all seasons. Advanced oxidation makes that consistency achievable, and nutrient recovery turns the cost of treatment into a revenue stream that compounds over every production cycle.
Conclusion
To summarize, seasonal organic load spikes expose a predictable, chemistry-driven failure mode for fixed chlorine systems. Ozone relies on direct molecular fragmentation and indirect hydroxyl radical mineralization, and these dual oxidation pathways provide inherent adaptability that eliminates this vulnerability. As regulatory standards tighten, modern facilities abandon outdated metrics and design wastewater treatment processes for seasonal peaks as baseline operating conditions.
Properly engineered advanced oxidation systems represent essential infrastructure to navigate volatile environments. Reliable treatment and reuse management ensures long-term operational stability. Agritopia develops precise ozone technologies that adapt to seasonal load fluctuations. Reach our for consultation to see how our technologies can help you meet your specific treatment requirements.






