Introduction to Drinking Water Standards
Facility managers face new challenges as regulatory frameworks tighten limits on emerging contaminants, disinfection byproducts, and resistant pathogens. They relied on conventional chlorination as the foundation of municipal water purification for over a century, but they increasingly struggle to satisfy strict safety mandates because chlorination generates prohibited chemical byproducts.
Compliance expectations shifted globally when the U.S. Environmental Protection Agency issued enforceable PFAS drinking water standards in April 2024. The European Union followed with its own strict PFAS limits that took effect in 2026. This regulatory shift forces facility operators to reconsider their primary infrastructure investments and evaluate unified water treatment platforms that address multiple contaminant classes at once.
Ozone systems represent a practical solution to this challenge because ozone relies on a single oxidation process to cover several regulatory categories simultaneously.
How Global Regulatory Frameworks Tighten Drinking Water Standards
Drinking water standards define the boundaries of acceptable water quality, and they now extend beyond basic microbial safety into strict chemical, physical, and radiological limits. Compliance frameworks enforce Maximum Contaminant Levels (MCLs) that set arsenic limits at 10 parts per billion and cap total trihalomethanes at 80 parts per billion. These thresholds reflect guideline values that the World Health Organization also endorses, and facilities that export products to international markets must satisfy the most restrictive standard in each contaminant category.
The gap between older limits and current requirements remains significant. Every water treatment procedure must now cover a broader contaminant spectrum because emerging threats such as PFAS, microplastics, and pharmaceutical residues create new regulatory pressures. Facilities that understood their obligations under previous frameworks face a fundamentally different compliance landscape today.
Environmental Forces Behind Stricter Drinking Water Standards
Environmental pressures explain why drinking water standards tighten on multiple fronts at the same time. Climate change degrades source water quality through drought cycles that concentrate contaminants. Extreme precipitation events overwhelm intake systems with turbidity and surface runoff, and elevated temperatures accelerate chemical reaction rates in distribution networks. The 2024 UN World Water Development Report frames water quality degradation as both an environmental and equity crisis, and confirms that communities with the fewest resources bear the greatest burden from these shifts.
Regulators respond to these pressures and move from voluntary guidance toward enforceable mandates. Government programs now provide technical assistance to small water systems that lack the engineering capacity to evaluate water treatment upgrades independently. This initiative signals a broader policy direction: regulators expect every facility to meet stricter standards regardless of system size, and they build support infrastructure to make that expectation realistic.
The Disinfection Byproduct Paradox in Conventional Water Treatment
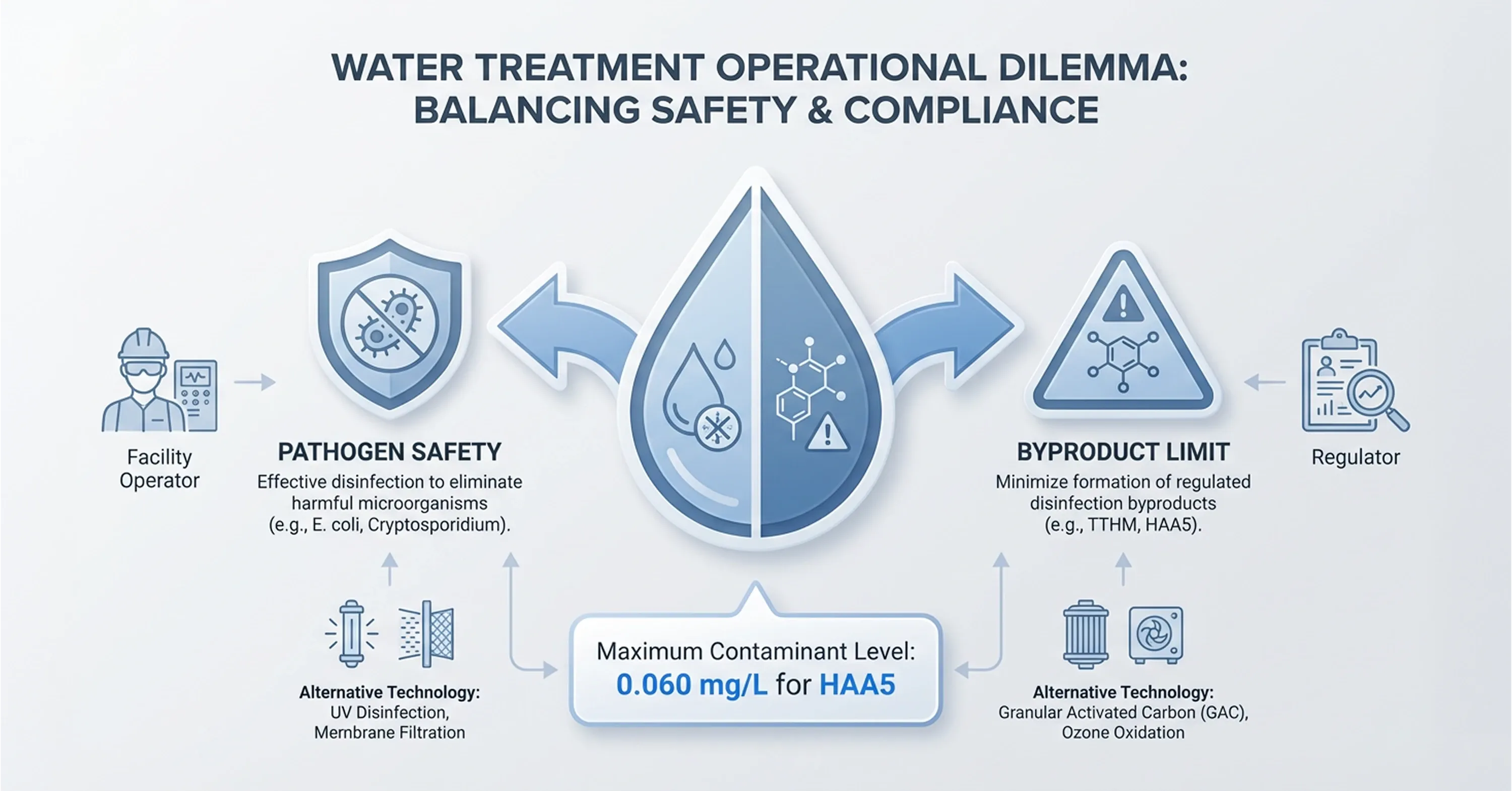
Conventional chlorination creates a regulatory contradiction that sits at the center of modern water treatment challenges. Chlorine reacts with naturally occurring organic matter in source water and generates two categories of harmful disinfection byproducts that regulators now tightly control.
-
Total trihalomethanes represent the first category.
-
Haloacetic acids represent the second category. The Maximum Contaminant Level for five regulated haloacetic acids stands at 0.060 milligrams per liter, also enforced as a running annual average that adds further compliance layers for facility operators.
This paradox presents a straightforward challenge. Facility operators raise byproduct concentrations toward or beyond regulatory ceilings when they increase chlorine doses to meet microbial safety targets. They risk inadequate pathogen inactivation when they reduce chlorine to control byproducts. Utilities that rely solely on chlorination find themselves trapped between two sets of regulations that pull in opposite directions.
This tension undermines the reliability of single-disinfectant strategies and explains why regulatory bodies increasingly encourage multi-barrier approaches. The safety margins that once existed under older byproduct limits have narrowed significantly. Facilities adopt alternative oxidation technologies like ozone because operational adjustments alone cannot resolve this conflict.
How Ozone Systems Address Multiple Regulatory Requirements
Ozone possesses chemical properties that allow a single platforms to address regulatory requirements that would otherwise demand multiple separate systems. Ozone delivers roughly twice the oxidizing power of chlorine with an oxidation potential of 2.07 volts.
This elevated reactivity means ozone reacts up to 3,000 times faster than chlorine to neutralize microorganisms, break down organic contaminants, and degrade complex molecular structures. Ozone also features a half-life of 15 to 20 minutes and decomposes into molecular oxygen instead of persistent chemical residuals in treated water.
This combination of high reactivity and rapid decomposition translates into concrete advantages across multiple regulatory categories. Ozone treats chlorine-resistant organisms, avoids the generation of halogenated byproducts, and initiates the degradation of emerging contaminants within the same procedure. Advanced oxidation processes pair ozone with hydrogen peroxide or ultraviolet light to extend this capability further through hydroxyl radicals that attack even the most persistent molecular bonds.
How Ozone Addresses Chlorine-Resistant Pathogens
Cryptosporidium parvum oocysts represent one of the most stubborn challenges in treatment facilities because their thick cell walls resist chlorine at concentrations normally used in municipal systems. Ozone's high oxidation potential breaches these cell walls effectively. A study published in Water Research demonstrated that ozone treatment effectively managed viral loads in conventional sewage treatment and confirmed ozone's broad-spectrum efficacy against organisms that survive chlorination.
This capability builds trust in ozone as a primary disinfection barrier for facilities that draw from surface water sources. Cryptosporidium contamination events in these sources pose the greatest public health risk. Chlorine-based systems typically require extended contact times to achieve equivalent pathogen reduction, but ozone achieves this reduction directly and avoids the formation of harmful chemical byproducts.
How Ozone Minimizes Byproducts
Ozone avoids the fundamental chemical reaction that creates regulated halogenated byproducts. Because ozone does not contain chlorine atoms, it cannot form trihalomethanes or haloacetic acids when it contacts organic matter in source water. This elimination removes the regulatory paradox that traps chlorine-dependent utilities and provides immediate compliance headroom.
Ozone reverts to molecular oxygen once it completes its oxidation reactions. This decomposition pathway leaves no persistent chemical residual that could react further downstream in distribution systems. The stability of treated water improves because the disinfectant itself does not become a source of secondary contamination. Facilities that operate near their byproduct compliance ceiling gain regulatory relief when they replace or supplement chlorination with ozone.
Ozone's Role in Emerging Contaminant Water Treatment
Emerging contaminant regulations represent the next frontier of compliance pressure. Ozone degrades microplastic particles by breaking polymer chains at the molecular level and achieves dramatically higher mass reduction rates than chlorine under identical conditions. This capability matters because regulators expect to formalize microplastic limits in the late 2020s as monitoring data matures.
Ozone also serves a strategic role in multi-barrier water treatment trains for per- and polyfluoroalkyl substances (PFAS). Ozone alone does not fully mineralize these compounds, but it removes competing organic contaminants from source water. This removal improves the efficiency of downstream granular activated carbon filters designed specifically for PFAS capture. This pre-treatment function extends filter bed life and reduces operating costs, which makes the compliance strategy economically sustainable.
Transparent Bromate Management in Ozone-Based Systems
An honest assessment of ozone's limitations strengthens the case for informed adoption. When ozone oxidizes naturally occurring bromide ions in source water, it produces bromate, a regulated contaminant with its own drinking water standards ceiling. Regulatory frameworks set the Maximum Contaminant Level for bromate at 0.010 mg/L for systems that use ozone disinfection. This standard requires monthly sampling, annual average calculations, and mandatory violation reporting within 30 days.
Research published in ACS Environmental Science and Technology confirms that bromate formation depends on both bromide content and ozone mass transfer design. This means system engineering choices directly control regulatory exposure. Operators minimize bromate through several proven strategies:
-
They maintain pH depression below 6.5 during ozonation to shift the reaction pathway away from bromate formation.
-
They calibrate controlled ozone dosing to source water bromide levels and apply only the precision needed for target contaminant destruction.
-
They use sequential treatment approaches where ozone handles specific contaminant removal while alternative disinfectants provide residual microbiological protection downstream.
These operational controls do not eliminate the challenge, but they reduce it to a manageable engineering problem. Facilities that monitor bromide levels in their source water and adjust dosing protocols accordingly maintain compliance with bromate limits and preserve ozone's broader advantages.
How Drinking Water Standards Extend to Agricultural Facilities
Drinking water standards increasingly shape commercial agriculture as food safety regulations adopt equivalent microbial and chemical thresholds for water that contacts edible crops. Ozone's dual functionality in commercial food production extends its compliance value well beyond municipal applications.
In recirculating hydroponic systems, ozone treats nutrient solutions for bacteria and viruses that cause crop losses from pathogens like Phytophthora, Pythium, and Fusarium. Because ozone decomposes into molecular oxygen after it completes its oxidation reactions, it simultaneously addresses root-zone hypoxia, the primary physiological stressor in intensive hydroponic crops. This oxygen enrichment improves nutrient uptake and plant vigor through the same treatment step that removes pathogens.
The safety and reliability gains compound when ozone integrates with precision irrigation technology that employs sensors and automated scheduling. Ozone-treated water delivered through smart micro-irrigation systems ensures microbial compliance at the point of crop contact and optimizes dissolved oxygen levels in the root zone. The result is a single treatment investment that satisfies food safety regulators, reduces crop disease pressure, and conserves water through recirculation rather than discharge.
Infrastructure Preparation for Future Regulatory Deadlines
Public water systems face strict regulatory timelines for infrastructure decisions. Regulatory frameworks require initial PFAS monitoring in the near term. These requirements apply regardless of system size, and they arrive against a backdrop of expanding contaminant lists that grow as monitoring data matures and microplastic regulations crystallize.
Compliance pressure now extends beyond water chemistry into operational security. Regulatory bodies have adopted cybersecurity requirements for water and wastewater facilities. These rules add digital protection standards to the already complex regulatory environment, and every water treatment procedure must now account for both chemical performance and secure operational controls.
Ozone's broad-spectrum oxidation capacity offers flexibility against this expanding regulatory landscape. A treatment platform that already addresses microbial safety, disinfection byproducts, and emerging contaminant pre-treatment does not require wholesale replacement when regulators add new compounds to their watchlists. This certainty matters because oxidation infrastructure covers current mandates and anticipated trajectories simultaneously.
Conclusion
To summarize, facility operators must evaluate their infrastructure decisions carefully because drinking water standards continue to tighten around emerging contaminants and disinfection byproducts. Utilities face strict monitoring deadlines through the end of the decade, and this trajectory makes conventional single-target water treatment approaches increasingly difficult to sustain.
Ozone-based systems provide operators with a clear pathway to achieve compliance across multiple regulatory frameworks at once. Ozone technology treats resistant pathogens, minimizes chemical byproducts, and degrades complex contaminants through a unified oxidation process.
Agritopia provides smart irrigation, fertigation controllers, and advanced water purification systems that help facilities align their operations with strict compliance requirements. To explore how ozone-based solutions can support your facility's infrastructure decisions, contact us for a consultation.






